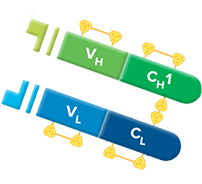
The general features of antibodies described below will focus on the IgG1 framework. The other isotypes are monomeric (a monomer is defined here as a pair of HC-LCs.). The IgA and IgM isotopes have an additional J-chain, which allows the formation of dimers and pentamers, respectively. IgEs and IgMs have one variable and four constant domains. IgAs, IgDs, and IgGs have three constant (C) and one variable (V) domains. In comparison, human antibody HCs can be one of five isotypes, IgA, IgD, IgE, IgG, and IgM, each with an independent role in the adaptive immune system. Both LC classes have two domains, a constant domain (CL) and a variable domain (VL). Human LCs can be one of two functionally similar classes, κ or λ. The two HCs of the heterotetramer are also linked by disulfide bridges. The HC and LC of the heterodimer are linked through disulfide bonds. In natural systems, the pairing of one LC with one HC associates with another identical heterodimer to form the intact immunoglobulin. Human immunoglobulins are Y-shaped proteins composed of two identical light chains (LCs) and two identical heavy chains (HCs). The overall structure of antibodies, including the folding pattern of the individual domains and basic features of the antigen-combining sites, has been the subject of several reviews. The structural data includes complexes of these molecules with proteins, other macromolecules, peptides, and haptens. At present, the Protein Data Bank (PDB) contains over 3500 structures of antibody fragments (Fabs, Fvs, scFvs, and Fcs), as well as a small number of intact antibody structures. Our knowledge of the three-dimensional structure of antibodies has emerged from crystallographic studies reported from numerous laboratories beginning in the 1970s. The engineering approaches being used are based on our knowledge of protein structure and, in particular, our knowledge of how the structures are linked to their function. Different strategies of preparing bispecific and multispecific molecules for an array of therapeutic applications are included.Ĭurrently, all antibodies and antibody-derived macromolecules being developed for a wide spectrum of therapeutic indications require protein engineering. 
We also review the design and selection of binding arms, and avidity modulation. The platforms examined include the development of antibodies, antibody fragments, bispecific antibody, and antibody fusion products, whose efficacy and manufacturability can be improved via humanization, affinity modulation, and stability enhancement. In this review, our basic understanding of the antibody structure is described along with how that knowledge has leveraged the engineering of antibody and antibody-related therapeutics having the appropriate antigen affinity, effector function, and biophysical properties. Our knowledge of the structure–function relationships of antibodies provides a platform for protein engineering that has been exploited to generate a wide range of biologics for a host of therapeutic indications. 1994.Antibodies and antibody-derived macromolecules have established themselves as the mainstay in protein-based therapeutic molecules (biologics). The use of monovalent Fab fragments avoids this problem. Capture of the primary antibody would allow detection of that primary by a labeled secondary antibody, resulting in background staining or apparent signal overlap. After binding endogenous IgG or the first primary antibody, some antigen binding sites on a divalent secondary antibody may remain unoccupied, which could capture a primary antibody introduced in a subsequent step. Why is Monovalency Important?ĭivalent (whole IgG or F(ab') 2 fragment) antibodies are not recommended for blocking because they have two antigen binding sites. they are monovalent), and they are non-precipitating. They can be used for these purposes because each Fab fragment has only a single antigen binding site (i.e. Monovalent Fab fragments of affinity-purified secondary antibodies are offered to block endogenous immunoglobulins in tissue sections or on cell surfaces, to cover (block) immunoglobulins when double labeling primary antibodies from the same host species, or to Fab-label primary antibodies prior to incubation with the experimental sample. Label primary antibodies in solution as an alternative to chemical conjugation with FabuLight™ antibodies.Double labeling primary antibodies from the same host species.Blocking Endogenous Immunoglobulins to reduce background staining.View Fab Fragment Generation by Papain Digestion Fab Fragments Can Be Utilized in Three Key Ways:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
